Academic team: Michel L. Tremblay, PhD. & David Langlais, PhD. (McGill University)
Receptor organization team: Pierre Laneuville, MD. and Linda Peltier, PhD. (McGill University Health Centre) and Kanyr Pharma Inc.
Partners organizations: The Rosalind and Morris Goodman Cancer Institute and the Richard and Edith Strauss Foundation
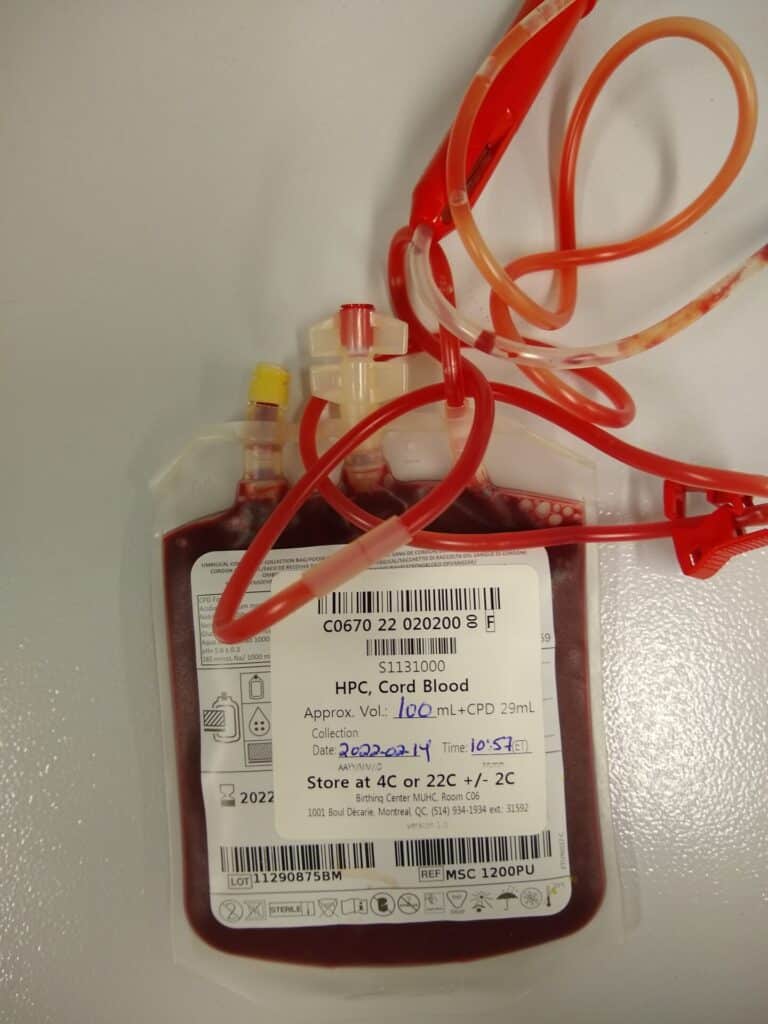
Cancer immunotherapies, such as modified T-cells with Chimeric Antigen Receptors (CAR-T), can elicit strong responses and even cure some blood cancers such as acute myeloid leukemia (AML). However, CAR-T cell drawbacks include poorer responses against solid tumours, autologous cell requirements, high manufacturing costs, and potentially fatal toxicities. These limitations can be overcome by natural killer (NK) cells. These innate immune cells can efficiently kill cancer cells, allowing the development of non-genetically modified NK cells to treat both blood and solid tumour cancers. The anti-tumour activity and response to stimulation of primary cord blood-derived NKs (CB-NK) are superior to those of NK cell lines, and are being used in clinical trials in the U.S. and China.
This project will use genomics solutions to improve the clinical management of hard-to-treat cancers like AML through CB-NK cell therapy. It will establish a comprehensive resource for “off-the-shelf” NK cell immunotherapy. Its unique methodologies will enable the processing, storage and recovery of CB-NK cells; optimisation and simplification of their clinical use; and national distribution of the therapeutic CB-NK cell bank to treat a larger cohort of cancer patients. The project team will develop the protocol/documentation for Health Canada approval of phase I trials to bring these therapies to the clinic, followed by a multi-institutional clinical trial with the Canadian Cancer Trial Group.
The acceleration of CB-NK cell therapies will place Canada at the forefront of this fast-moving and strategically important field. Canadian patients will benefit from safer, more effective immunotherapies with the potential to tackle diverse hard-to-treat cancers, while their “off-the-shelf” nature will significantly lower healthcare costs. Economic benefits include increased revenues associated with licensing and development of commercial products based on the project’s IP, as well as economic growth and job creation due to innovation in the cell therapy sector, where the global market is growing rapidly.